Type-2 Diabetes
FGF-1 for Type-2 Diabetes:
Zhittya Genesis Medicine is developing a drug treatment, which we believe can effectively cure Type-2 Diabetes.
Leading medical research institutions and Zhittya believe that Type-2 Diabetes is a neurological disease caused by the glucose sensing neurons in the brain not performing normally. We believe, our molecule, FGF-1, resets the glucose-sensing neurons in the brain.
Starting in 2014, medical research from leading institutions like the Salk Institute, Howard Hughes, Scripps, Yale University, and the Health & Human Services (HHS) showed that FGF-1 reverses Type-2 Diabetes in diabetic animal models. It boosts insulin sensitivity in muscles and reduces liver glucose production, all without risks like low blood sugar, weight gain, or liver damage. Studies were repeated year after year demonstrating this huge potential to effectively cure diabetes.
Zhittya Genesis Medicine is advancing this potential treatment for Type-2 Diabetes. In the British Virgin Islands, where Zhittya is cleared to do a medical research study, Zhittya Genesis Medicine has dosed 25 patients suffering from Type-2 Diabetes. Over the course of the study these patients were no longer diabetic. The longest has remained diabetes-free for 2 years.
Medical Research Study to Reverse Type-2 Diabetes- Las Vegas, NV
Zhittya Genesis Medicine has received Institutional Review Board (IRB) approval to do a 100 person medical research study in Las Vegas, Nevada to confirm whether FGF-1 can reverse and/or effectively cure Type-2 Diabetes.

This is a sample of one of the many publications, this one from the Health & Human Services showing that FGF-1 can reverse and effectively cure Type-2 Diabetes. For more of these papers visit our main website: zgm.care
Register for Medical Research Study to Reverse Type-2 Diabetes. Located in: Las Vegas, Nevada
If you have Type-2 Diabetes and are interested in potentially participating in our medical research study to reverse Type-2 Diabetes, complete the form below to express your interest. For those patients that are selected to participate, they will bear no cost for participating in the study other than their own travel and accommodations. This is a voluntary medical research study to evaluate FGF-1's ability to potentially reverse Type-2 Diabetes. We'll screen applicants for eligibility, but there's no guarantee of participation or specific results. All processes follow ethical guidelines, including informed consent.
Disclaimer: This form is for expressing interest in a medical research study only. It does not constitute enrollment, medical advice, or a guarantee of participation. All studies comply with FDA guidelines and ethical standards. Consult your physician before considering any research involvement.
Zhittya is contracting a fund of $5 million USD to fund this study to verify if FGF-1 can reverse Type-2 Diabetes. Investors interested in funding the study have the potential profit returns of:
10x return from a 5% royalty on all FGF-1 revenues for Type-2 Diabetes OR
2x their investment (at investor put option) after 48 months, if the investor is not happy
Minimum: $25,000.
Submit the form for more details. This is informational only—not an offer to invest.
Information on Investing into Medical Research Studies for Type-2 Diabetes:
Disclaimer: This is not an offer to sell, nor a solicitation of an offer to buy, any securities. Any potential investment opportunities will be provided only to qualified, accredited investors in compliance with SEC regulations, including Rule 506(c) of Regulation D where applicable. Investments involve significant risk, including the potential loss of principal. Past performance is not indicative of future results. For details, accredited investors may request a private placement memorandum upon qualification. Zhittya Genesis Medicine makes no representations or warranties regarding returns or outcomes.
History
FGF-1 is a potent stimulator of angiogenesis, the growth of new blood vessels, and is capable of growing new blood vessels in ischemic areas of the body, including the brain. Research has indicated that a lack of blood perfusion to glucose-sensing neurons in the brain could lead to Type-2 Diabetes.
The management of Zhittya has spent over $190 million dollars to advance this molecule bringing it as far as US FDA Phase IIA/Phase IIB clinical trials in various indications.
In the past, FGF-1 has been able to grow new blood vessels in the human body. In a US FDA Phase IIA clinical trial, conducted at the University of Cincinnati, our drug was able to grow new blood vessels in the hearts of individuals with coronary artery disease, improving many of their symptoms. Moreover, this drug has been studied in diabetic foot uclers, stroke recovery, and more.
Watch as Zhittya Genesis Medicine’s drug is used to grow new blood vessels in the human heart in a US FDA Phase IIA Clinical Trial done at the University of Cincinnati, and reported by ABC Nightly News. Zhittya believes that by growing new blood vessels in the human brain, that the Type-2 Diabetes could be reversed if not effectively cured.
Watch our theory of how we believe FGF-1 can be used to treat neurodegenerative diseases by growing new blood vessels. In this animated video which shows our theory of our blood vessels grow, reestablishing blood flow to the brain.
News
-
![]()
January 2026 Update
A growing body of evidence suggests major medical breakthroughs could dramatically extend healthy human lifespan by addressing the root causes of disease. Intranasal FGF-1 continues to show strong results, with 90% of participants reversing type-2 diabetes within weeks and meaningful improvements observed in Parkinson’s disease. Heart studies are set to begin in The Bahamas for no-option patients, building on prior success in extending survival. International interest is accelerating through Nevada’s Freedom to Try initiative, creating new pathways for innovation that bypass decades-long regulatory delays and bring life-altering treatments directly to patients.
-
![]()
December 2025 Update
A major initiative is underway to establish a Nevada constitutional amendment granting patients and physicians the freedom to access advanced medical innovations without FDA barriers. The proposal aims to replace the failed Right to Try system, which has helped only 21 patients nationwide in seven years. The effort has already attracted strong interest from U.S. and international biotech companies seeking faster paths to real-world research. Alongside this initiative, progress continues in diabetes, Parkinson’s, stroke, and heart disease studies, with growing momentum toward patient-driven, market-based medical innovation.
-
![]()
November 2025 Update
FGF-1 continues to deliver strong human results across multiple diseases. In Las Vegas, early participants in the type-2 diabetes study are rapidly reducing or eliminating medication, while enrollment demand remains overwhelming. Parkinson’s patients report sustained improvements in mobility, cognition, speech, and daily function, with no adverse events observed across hundreds treated. Preparations are underway to launch studies for chronic wounds and no-option heart disease in early 2026. Despite long-standing regulatory barriers, growing public, patient, and investor support signals accelerating momentum toward treatments that address root causes rather than symptoms.
-
![]()
October 2025 Update
FGF-1 continues to show powerful results, with Zhittya’s type-2 diabetes study in Las Vegas revealing early patient successes—some reducing or eliminating medication within weeks. Global interest is surging, especially across the Arab Gulf and India, where diabetes rates are highest. Funding for the 100-person study is nearly complete, with chronic wound trials next. Parkinson’s patients also report major recoveries, including medication withdrawal and restored mobility. CEO Dan Montano celebrates growing investor support and patient breakthroughs, declaring that after 27 years, Zhittya’s mission to cure vascular and neurodegenerative diseases is entering its most successful phase.
-
![]()
September 2025 Update
A groundbreaking discovery—cellular rejuvenation through FGF-1—reveals that this molecule restores normal cell function by repairing intracellular dysfunction, offering promise for aging, diabetes, and neurodegenerative diseases. The 100-person U.S. study to reverse type-2 diabetes begins soon, following prior success with 25 diabetes-free patients in the British Virgin Islands. Funding is 70% complete, with a second chronic wound healing study launching in October. Zhittya is also expanding manufacturing in Reno and advancing research in Parkinson’s, stroke, dementia, and Alzheimer’s, marking a new era of cellular repair and regeneration.
-
![]()
August 2025 Update
Zhittya Genesis Medicine’s August 2025 update highlights progress in reversing type-2 diabetes, with 25 patients in the British Virgin Islands now diabetes-free and U.S. trials in Las Vegas set to begin. Over half of the $5 million funding goal is already secured.
The company is also preparing trials for chronic wound healing in October and advancing FGF-1 studies in Parkinson’s, stroke, dementia, and Alzheimer’s through higher dosing.
CEO Dan Montano criticizes regulatory delays that have cost millions of lives and calls for urgent action to bring FGF-1 therapies to patients suffering from heart disease, diabetes, and neurodegenerative conditions.
-
![We believe that Type-2 Diabetes is a Neurological disease]()
July 2025 Update
Zhittya Genesis Medicine’s July 2025 update focuses on the launch of a major U.S.-based Medical Research Study to reverse type-2 diabetes, following success with 20 patients in the British Virgin Islands. The study in Las Vegas will explore optimized dosing across 100 participants. Zhittya also announced plans to investigate type-1 diabetes after emerging research showed FGF-1 may regenerate insulin-producing beta cells.
Progress continues in neurodegenerative diseases like Parkinson’s and MSA, with ongoing intranasal FGF-1 dosing studies. Patients report notable recoveries in cognition, mobility, and quality of life.
Funding efforts for medical research studies have already secured over $2 million, with more R&D partnerships planned. Zhittya criticizes systemic barriers and calls out Big Pharma for ignoring FGF-1’s curative potential. CEO Dan Montano reaffirms the company’s mission to cure, not manage, disease—urging urgency and purpose to deliver life-saving therapies.
-
![June 2025 Update]()
June 2025 Update
Zhittya Genesis Medicine’s June 2025 update emphasizes the urgency of advancing medical breakthroughs during a critical 43-month window. Key successes include reversing type-2 diabetes in the British Virgin Islands and launching FDA-approved studies in Nevada with 100 participants.
The company also progresses in treating neurodegenerative diseases like Parkinson’s, stroke, and dementia, with life-changing recoveries and improved dosing strategies. Their "Swift 6" initiative targets six major diseases with plans to produce results within nine months.
Showcasing innovations at FreedomFest 2025, Zhittya strengthens partnerships and public awareness. Having invested 27 years and $190 million, they remain committed to overcoming hurdles to deliver FGF-1 therapies for vascular and neurological conditions affecting millions.
-
![May 2025 Update]()
May 2025 Update
Zhittya Genesis Medicine’s May 2025 update highlights breakthroughs in medical innovation. Celebrating three years of Parkinson’s treatments, Zhittya showcased remarkable recoveries, including wheelchair-bound patients walking again. CEO Dan Montano’s interview on the Wayne Allyn Root Show spotlighted successes in treating Parkinson’s, stroke, and type-2 diabetes.
The company launched U.S.-based Medical Research Studies to reverse type-2 diabetes and neuropathy, building on prior 100% diabetes reversals among ten patients. FDA changes may accelerate treatment approvals, potentially cutting years from timelines.
A new biomanufacturing lab in Reno, Nevada, scales production of FGF-1, critical for vascular regeneration, while research into Long COVID addresses lingering symptoms in millions. Montano calls for swift action to achieve breakthroughs within a pivotal 44-month window, aiming to transform care for vascular-related diseases and save millions of lives.
-
![Our April 2, 2025 Dinner presenting on our research for Type-2 Diabetes]()
April 2025 Update
Zhittya Genesis Medicine’s April 2025 update showcases groundbreaking advancements in FGF-1 therapy and advocacy for medical innovation. Dr. Jack Jacobs, President, was honored as Nevada Bio Organization’s Man of the Year for his 40-year career, notably treating over 200 Parkinson’s patients with significant recoveries and advancing FGF-1 for heart disease, stroke, dementia, Alzheimer’s, and type-2 diabetes. Zhittya’s research in the British Virgin Islands successfully reversed type-2 diabetes in 10 patients, highlighting the untapped potential of FGF-1, which remains stalled by regulatory barriers. The update calls for enhancing Nevada’s “Right to Try” law to spark an economic boom and accelerate access to treatments for neurodegenerative diseases, neuropathy, and stroke. CEO Dan Montano emphasizes a 45-month window to bypass FDA delays, rallying support to bring FGF-1’s life-saving potential to millions suffering from vascular-related diseases.
-
![Colored diagram of a human brain showing inner structures, including cross-sectional view of the brain with labeled regions.]()
March 2025 Update
Zhittya Genesis Medicine’s March 2025 update highlights an overwhelming response to their Washington University fMRI study, which showed a 21% increase in brain blood flow after FGF-1 treatment. This has sparked global interest, particularly in its potential for stroke recovery.
The 5-week Neuropathy Medical Research Study reports an 80% reduction in foot pain, with more data expected in the coming months. Zhittya also announced five pending patents covering intranasal and injected FGF-1 treatments for Parkinson’s, brain diseases, diabetes, lung conditions, and neuropathy.
Additionally, Dr. Jack Jacobs has been named NevBio’s Man of the Year, with an award presentation on April 2 in Las Vegas. CEO Dan Montano recently appeared on the Joe Cortez Show, and Zhittya is launching its own Medical Information Show. Meanwhile, their Outreach Program is expanding, with clinics, doctors, and patients advocating for FGF-1’s groundbreaking potential.
-
![A heatmap with orange and red colors showing two pixelated mouse-like shapes outlined in white, with green axes. Text at the bottom reads "6 Months After Dosing with FGF-1."]()
fMRI Update
Zhittya Genesis Medicine’s fMRI update showcases a major breakthrough in their ongoing Parkinson’s research. The 6-month follow-up fMRI scan of a treated patient revealed a 21.2% increase in blood flow to the substantia nigra, supporting their hypothesis that restoring vascular function can slow, stop, or even reverse neurodegeneration. The patient’s motor skills improved by over 50%, aligning with data from over 200 patients, who showed an average 54% motor skill improvement within six months. Additional patients are undergoing follow-up scans, further validating these findings. Zhittya continues to advocate for broader patient access to FGF-1 therapy, which may represent a disease-modifying breakthrough beyond symptom management. CEO Dan Montano highlights its potential in treating Parkinson’s, stroke, dementia, neuropathy, and lung diseases, marking a paradigm shift in neurodegenerative treatment.
-
![]()
February 2025 Update
Zhittya Genesis Medicine’s February 2025 update highlights significant strides in their ongoing research on FGF-1 therapy. The final data from their first Parkinson’s patient’s 6-month follow-up brain scan revealed a 21% increase in blood flow to the substantia nigra, reinforcing their hypothesis that vascular dysfunction underlies neurodegenerative diseases. Zhittya also initiated new medical research studies on neuropathy and respiratory (lung damage) conditions, expanding the potential applications of FGF-1 therapy. The update emphasizes the urgent need to modify the “Right to Try Law” to grant patients quicker access to innovative treatments. CEO Dan Montano underscores the transformative potential of FGF-1 to address not only Parkinson’s but also stroke, dementia, neuropathy, and lung diseases, expressing hope for broader acceptance and accelerated patient access in the near future
-
![January 2025 Update]()
January 2025 Update
Zhittya Genesis Medicine’s January 2025 update celebrates a major breakthrough in their Parkinson’s disease research. A functional MRI scan from one of their patients, Al, revealed a 42% increase in blood flow to the substantia nigra region of the brain six months after receiving FGF-1 treatment. This empirical data provides scientific validation of Zhittya’s long-held hypothesis that vascular dysfunction is the root cause of Parkinson’s and other neurodegenerative diseases. Encouraged by this milestone, Zhittya plans to expand MRI testing to more patients and accelerate efforts to bring FGF-1 to those suffering from conditions such as Alzheimer’s, stroke, and diabetes. The company remains committed to advancing clinical studies and advocating for regulatory changes to make their groundbreaking treatment accessible. CEO Dan Montano expresses gratitude for the patients participating in their studies and remains optimistic about the transformative potential of FGF-1 in the coming years
-
![]()
December 2024 Update
Zhittya Genesis Medicine’s December 2024 update emphasizes the promising progress of their FGF-1 treatment in reversing Parkinson’s disease and potentially addressing other neurodegenerative conditions. The update highlights the significant improvements seen in patients, with some regaining cognitive and motor functions, and the anticipation surrounding MRI scans at Washington University to confirm increased blood flow in the brain. CEO Dan Montano expresses optimism for 2025, envisioning it as a breakthrough year with expanded clinical studies in heart disease, diabetes, and neurodegenerative disorders. Zhittya is actively advocating for regulatory reforms to accelerate patient access to FGF-1 under the “Right to Try” law. As they push for FDA approvals and Pre-IPO funding, the company remains dedicated to providing life-changing treatments while navigating regulatory challenges
-
![]()
November 2024 Update
Zhittya Genesis Medicine's November 2024 update showcases advances in their FGF-1 treatment research, highlighting its potential to heal vascular dysfunction in the brain. With nearly 200 Parkinson’s patients treated, new protocols delivering 40 times the initial dose show promising results in motor and cognitive improvements. The update discusses using FGF-1 to treat neurodegenerative diseases like dementia, ALS, and autism, and its success in promoting neurogenesis. A key milestone includes upcoming brain scans at Washington University to measure improved blood flow in treated patients. Zhittya remains focused on pre-IPO funding to drive its FDA compliance and clinical trials in 2025.
-
![]()
October 2024 Update
Zhittya Genesis Medicine's October 2024 update highlights continued progress in treating neurodegenerative diseases. A notable case is shared involving a patient who, despite a severe stroke three days prior to treatment, experienced significant recovery over nine months, regaining mobility and cognitive abilities. CEO Dan Montano reflects on the uphill battle faced against established medical institutions resistant to change. The update underscores Zhittya’s hypothesis that Parkinson’s and similar diseases stem from vascular dysfunction. Dr. Jacobs’s presentation in Kuwait was met with positive interest, emphasizing the potential of FGF-1 treatment.
-
![]()
September 2024 Update
Zhittya Genesis Medicine's August 2024 update outlines their continued progress in treating Parkinson's disease and severe heart disease. In the British Virgin Islands, Cohort #27 for Parkinson's treatment began, with the company having now treated 180 patients with no significant adverse events. The update highlights improved motor and mental skills in patients and discusses plans to refine dosing protocols for better outcomes. Zhittya also made advancements in heart disease treatment, obtaining clearance in The Bahamas and receiving an overwhelming response from patients. Additionally, Dr. Jacobs is scheduled to present in Kuwait on Parkinson's treatments in October, further expanding Zhittya’s global presence. The company continues to seek pre-IPO investment to support these initiatives.
-
![]()
August 2024 Update
Zhittya Genesis Medicine's August 2024 update outlines their continued progress in treating Parkinson's disease and severe heart disease. In the British Virgin Islands, Cohort #27 for Parkinson's treatment began, with the company having now treated 180 patients with no significant adverse events. The update highlights improved motor and mental skills in patients and discusses plans to refine dosing protocols for better outcomes. Zhittya also made advancements in heart disease treatment, obtaining clearance in The Bahamas and receiving an overwhelming response from patients. Additionally, Dr. Jacobs is scheduled to present in Kuwait on Parkinson's treatments in October, further expanding Zhittya’s global presence. The company continues to seek pre-IPO investment to support these initiatives.
-
![]()
July 2024 Update
Zhittya Genesis Medicine's June 2024 update highlights their significant advancements in medical research. The company has received regulatory clearance to begin studies for severe heart disease in The Bahamas, Parkinson's disease in The Bahamas, and critical limb ischemia in the British Virgin Islands. Their recent trip to the British Virgin Islands marked the dosing of their 175th Parkinson’s patient with FGF-1, showing promising results. Upcoming initiatives include presentations in Armenia and a strong presence at FreedomFest in Las Vegas. Zhittya is also seeking pre-IPO investment to fund these expanding operations and research efforts. CEO Dan Montano underscores the urgency of advancing treatments, particularly for heart disease, the leading cause of death globally.
-
![A healthcare worker and an elderly man smiling and holding hands in a medical setting.]()
June 2024 Update
Zhittya Genesis Medicine's June 2024 update highlights their significant advancements in medical research. The company has received regulatory clearance to begin studies for severe heart disease in The Bahamas, Parkinson's disease in The Bahamas, and critical limb ischemia in the British Virgin Islands. Their recent trip to the British Virgin Islands marked the dosing of their 175th Parkinson’s patient with FGF-1, showing promising results. Upcoming initiatives include presentations in Armenia and a strong presence at FreedomFest in Las Vegas. Zhittya is also seeking pre-IPO investment to fund these expanding operations and research efforts. CEO Dan Montano underscores the urgency of advancing treatments, particularly for heart disease, the leading cause of death globally.
-
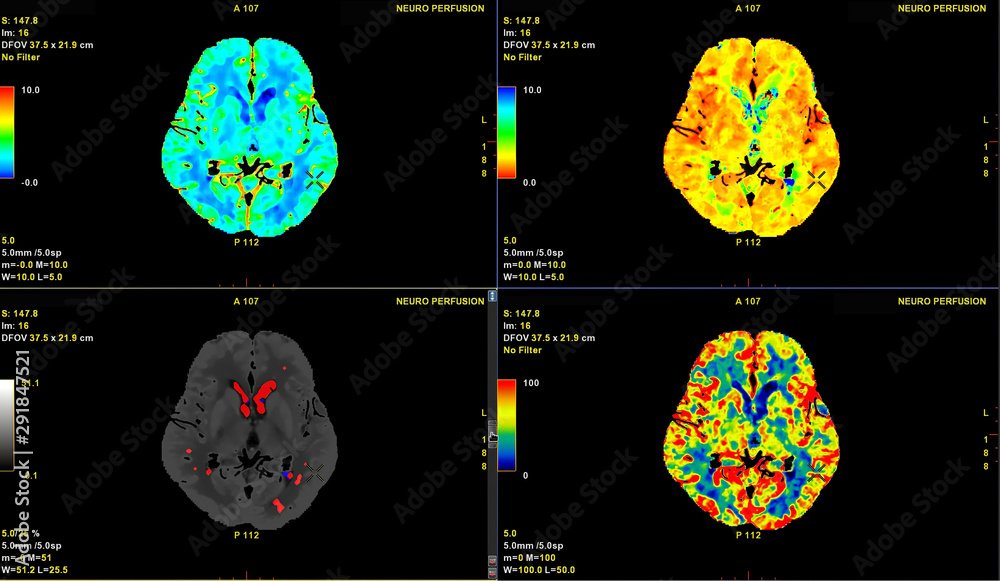
![Four brain scan images showing different color-coded views of neural activity in a human brain, with labels indicating neuroperfusion measurements and color scales ranging from blue to red.]()
May 2024 Update
Zhittya Genesis Medicine’s latest update provides an insightful overview of their ongoing efforts and groundbreaking research in 2024. The company has achieved significant milestones, including the first brain scan measuring blood perfusion in a Parkinson’s patient and a successful spinal tap delivery of FGF-1. Their recent conference in The Bahamas highlighted potential treatments for Parkinson’s and coronary heart disease, and they are planning new applications for critical leg ischemia and diabetic foot ulcers. Celebrating two years of treating Parkinson's patients, Zhittya has treated over 150 subjects and continues to work with US agencies to seek approval for their innovative treatments.
-
![Aerial view of a tropical beach with white sand, turquoise water, and beachfront houses surrounded by lush greenery.]()
Zhittya Research Study Approved in The Bahamas!
Zhittya Genesis Medicine has obtained regulatory clearance to begin conducting research studies in The Bahamas. Zhittya Genesis Medicine will begin utilizing FGF-1 in patients suffering from Parkinson’s disease utilizing an intranasal delivery method. Zhittya plan to begin these studies in the coming months.
-
![View of a swimming pool overlooking the ocean, with lounge chairs, potted plants, and a white fence in a tropical setting.]()
-
![A group of around 25 people posing for a photo inside a covered area with large windows showing a scenic ocean view in the background. The group is smiling and appears happy, dressed in casual and semi-formal clothing. Some are seated, and others are standing.]()
Zhittya Doses 150th Patient
April 2024:
Zhittya Genesis Medicine is proud to announce that they have dosed their 150th Parkinson’s Disease patient in their medical research studies, proving as a substantial milestone for Zhittya’s potential treatment. With 150 patients now dosed, Zhittya has seen no reported adverse events and a 53% improvement, on average, in reported motor scores. -
![]()
April 2024 Update
This update from Zhittya Genesis Medicine, dated April 2024, highlights their continued efforts in advancing treatments for Parkinson’s disease and other health conditions using FGF-1. The company celebrates treating over 150 Parkinson's disease sufferers with notable improvements and minimal side effects. As the two-year mark approaches since the first human dosing, they are beginning to gather long-term efficacy data. Furthermore, the update contrasts FGF-1 treatment with deep brain stimulation (DBS), emphasizing FGF-1's non-invasive application and broader health benefits over the surgical and potentially riskier DBS.
-
![A female scientist wearing safety goggles and blue gloves working with a pipette in a laboratory.]()
-
![Illustration of neurons with their cell bodies and long, branching dendrites, connected to a blood vessel.]()
March 2024 Update
Zhittya Genesis Medicine’s March 2024 update discusses their recent activities, particularly focusing on the treatment of Parkinson's disease patients with FGF-1 through their ongoing medical research studies. The company's ongoing research is exploring how continual dosing with FGF-1 can aid in the recovery and improvement of patients' conditions. Despite the uncertainties and the complex journey of discovery, positive changes in patients have been observed, leading to renewed hope and potential treatment strategies. Additionally, the update delves into the intriguing aspect of where the administered FGF-1 goes within the brain, considering the vast number of molecules unaccounted for and the possible extensive repair work being undertaken within the brain's complex structure.
-
![Night view of Dubai's downtown skyline with illuminated skyscrapers and reflections on water.]()
February 2024 Update
Zhittya Genesis Medicine’s February 2024 update details the company's busy start to the year, with significant developments in their research and treatment offerings, particularly focusing on FGF-1's potential in addressing various diseases. Zhittya recounts successful presentations and interactions at the Dubai Cardiovascular Conference, emphasizing the international medical community's growing interest in FGF-1 for treating small vessel heart disease, Parkinson’s disease, and other neurodegenerative conditions. Additionally, it highlights the progress in brain scanning and blood flow imaging studies, aiming to empirically demonstrate FGF-1's effectiveness in improving cerebral blood perfusion.
-
![A nurse wearing a face mask and white uniform stands beside a patient lying on a hospital bed, preparing to perform a medical scan inside a large medical imaging machine.]()
January 2024 Update
Zhittya Genesis Medicine’s January 2024 update provides an insightful overview of their ongoing efforts and groundbreaking research in 2024. The company is making strides in treating Parkinson's disease especially as they work to develop strategic collaborations with renowned medical imaging centers in the United States and Italy. These partnerships aim to scientifically demonstrate how FGF-1 enhances brain blood flow and potentially reverses disease symptoms.
Meet Zhittya’s Team
-
![A man with glasses and gray hair wearing a suit and red tie, standing in front of a bookshelf filled with books.]()
Dan Montano
Chief Executive Officer
-
![An older man with white hair and glasses, wearing a white shirt and blue tie, against a blue sky with some clouds.]()
Dr. Jack Jacobs
President and Chief Science Officer
-
![A woman with long dark hair wearing a red dress, smiling slightly against a plain background.]()
Viktoriya Tamlenova-Montano
Executive Vice President
-
![Close-up of smiling older woman with short, blonde hair, wearing an orange top and a silver necklace against a plain gray background.]()
Judith Luell
Vice President of Operations
-
![Professional headshot of a smiling woman with short brown hair, glasses, and a black blazer over a white shirt, against a dark gray background.]()
Megan Kruitbosch
Research Scientist
-
![A professional headshot of a man wearing a dark suit, light shirt, and striped tie, smiling slightly against a dark background.]()
Tezo Albarran Martinez
Research Scientist
-
![Young man in a dark suit and red tie smiling in a modern office lobby.]()
Daniel Montano
Vice President of Drug Development
-
![A young man in formal attire standing outdoors on a sunny day with a building and fountain in the background.]()
Khiry Anderson
Technology and Marketing Manager
-
![A young man in a navy puffer jacket standing outdoors with a city skyline of tall skyscrapers behind him, near a river.]()
Sergiy Prokofiy Montano
Vice President of Marketing
Our Partners
-
![Logo for Cross Border Medical Tourism featuring a purple airplane circling a globe with latitude and longitude lines, over a gray background, with the text 'CROSS BORDER' in bold black and 'MEDICAL TOURISM' beneath.]()
Cross Border Medical Tourism
Zhittya Genesis Medicine’s partner in patient facilitation, patient logistics, and operations based in Los Angeles, California. Cross Border Medical Tourism ensures that all of our patients are cared for throughout our programs.
-
![Text that reads 'WundHealing' with 'Wund' in red and 'Healing' in blue.]()
WundHealing Biopharmaceuticals
Zhittya Genesis Medicine’s marketing partner based in Las Vegas, Nevada, helping Zhittya Genesis Medicine market and distribute our potential treatment following anticipated approval by the US FDA and other regulatory bodies.